Medical-Grade Clinical Research Translation Precision in the Age of AI
Protect Data Integrity and Patient Safety in Your Global Trials
In clinical research, inaccurate translation is a catastrophic risk to patient safety, data integrity, and regulatory approval. Language Scientific provides the medical-grade precision and regulatory understanding necessary to avoid the high costs of translation errors, ensuring your multi-national trials stay on track.




Why Generic AI Translation is a Risk to Your Clinical Trial
While AI offers efficiency, generic machine translations lack the contextual relevancy and scientific terminology required for complex clinical protocols. Language Scientific solves the “colossal need” for formal certification of accuracy, regardless of how the translation was generated.
The Risks We Eliminate for CROs:
- Patient Safety: Preventing life-altering errors in Informed Consent Forms (ICFs) and dosage instructions.
- Regulatory Delays: Avoiding costly setbacks in global trial execution due to non-compliant documentation.
- Data Integrity: Ensuring statistically relevant and accurate data for global regulatory submissions.
By implementing independent auditing and certification protocols, we act as a critical medical-grade guard-rail, validating the precision of every document to ensure total compliance and patient safety in a high-stakes clinical context.
Translation Capabilities
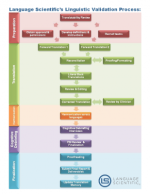
Linguistic Validation
Language Scientific’s Linguistic Validation group specializes in the translation and adaptation of Clinical Outcome Assessments in all therapeutic areas. We are industry experts in linguistic validation and our process has been designed to ensure that your eCOA is both culturally appropriate and conceptually equivalent in over 215 languages.
- Accurate Patient Insights: Ensure your clinical research captures meaningful, culturally appropriate patient data with expert linguistic validation services.
- Seamless Compliance: Meet global regulatory standards with translations tailored for patient-reported outcomes (PROs) and clinical questionnaires.
- End-to-End Support: From translation to cognitive debriefing, our team ensures every step aligns with your study’s goals.

DICOM Translation
Language Scientific understands the complexity and magnitude of DICOM files and has the tools, processes, technology and expertise to seamlessly redact and translate DICOM files accurately and expeditiously.
- Streamlined Imaging Data: Expertly translate DICOM files to ensure seamless communication and interpretation across global clinical trials.
- Precision in Patient Safety: Maintain data accuracy and regulatory compliance with our specialized medical translation expertise.
- Global Collaboration Ready: Enable effective cross-border collaboration with clear, localized imaging data for every stakeholder.

Endpoint Adjudication
Streamline your endpoint adjudication process with expert translation services tailored for global clinical trials. Our specialized solutions ensure every document and communication is clear, accurate, and compliant across all regions.
- Clarity for Adjudication Panels: Deliver precise translations of clinical data to support informed decision-making during endpoint reviews.
- Regulatory Confidence: Ensure compliance with international standards for endpoint adjudication documentation.
- Tailored Expertise: Work with industry specialists who understand the nuances of adjudication processes and medical terminology.

Patient Data Translation
Unlock the full potential of your patient data with expert translation services designed for global healthcare and research. Our solutions ensure your data remains accurate, compliant, and actionable across borders.
- Data Integrity Across Languages: Preserve the accuracy and clarity of patient data with expert translations for healthcare and clinical use.
- Global Compliance Made Easy: Meet international regulatory requirements with translations tailored to your specific data formats and needs.
- Specialized Expertise: Leverage our deep knowledge in life sciences to ensure your patient data supports effective decision-making worldwide.

The Only Solution Built for Clinical Research Precision
Language Scientific is the first and only translation company with a deeply experienced team focused exclusively on the scientific and medical fields.
- The Scientist-to-Scientist Advantage: We don’t just hire linguists; we hire peers. Every clinical project is handled by a dual-review team featuring certified linguists and a Subject Matter Expert (SME) with an advanced degree (MD or PhD) in your specific therapeutic area.
- The LS Certified™ Stamp: We provide the formal certification required to confirm medical-grade accuracy and compliance with global regulatory rules, giving you and your sponsors peace of mind.
- Fit-for-Purpose AI Workflows: We meet you where your needs are, offering AI-optimized, human-verified workflows that drive efficiency without compromising clinical quality.
Language Scientific ensures that these three steps ensure that the right guard-rails are in place for any translation we conduct, especially in the world of AI.
A large portion of the communication in the clinical trial process takes place between clinicians and patients. Depending on the study site and the level of literacy of the patients, the patient-facing materials must be translated to match their reading levels, and the cultural component is equally important. For instance, in a recent questionnaire, a patient in rural India was asked,
Specialized Services for Global Clinical Execution
We manage the entire translation process so you can focus on developing life-saving medical technologies. Our clinical expertise covers:
- Protocol & Investigator Brochures: Precise translation to ensure international sites follow identical study parameters.
- Informed Consent Forms (ICFs): Culturally sensitive and linguistically accurate documents that meet strict regulatory standards.
- Patient-Facing Materials: Adapting Patient Diaries, Questionnaires, and Recruitment materials to ensure comprehension and retention.
- Pharmacovigilance & Safety: Rapid, accurate translation of Adverse Event Reports (SAEs) and SUSARs.
Sample Clinical Documents We Translate
- Adverse Event Source Document
- Case Report Forms (CRFs)
- Data Sheets
- Development Safety Update Reports (DSURs)
- Dossiers
- Drug Registration Documentation
- Endpoint Adjudication Documents
- Informed Consent Forms
- Insert Leaflets
- Instructions For Use (IFUs)
- Marketing Collateral
- Master Batch Records
- Package Inserts and Labels
- Patient Diaries
- Patient Recruitment Materials
- Patient Reported Outcome Measures (PROMs)
- Patient Source Documents, Admission and Discharge, Labs
- Pharmacological Studies
- Pharmacovigilance and Postmarketing Surveillance
- Product Labels
- QoL Scales
- Regulatory Documents
- Scientific Papers
- Site Operation Manuals
- Study Protocols
- Suspected Unexpected Serious Adverse Reactions (SUSARs)
Trust and Reliability: The Four Pillars of Language Scientific
We provide transparency and reliability that satisfy both procurement and aggressive clinical timelines:
- 99.7% On-Time Delivery Guarantee: We understand that clinical trial timelines are absolute. We guarantee on-time delivery for every project.
- Full Transparency via LS Client Portal: Gain proactive access to project progress and documented quality checks for total oversight.
- Compliance-First Security: Our Quality Management System is ISO 17100 and ISO 9001 certified, with strict HIPAA and GDPR-compliant protocols to protect sensitive patient data.
4. Global Regulatory Mastery: Deep knowledge of country-specific requirements to prevent trial slowdowns.
Ready to Guard Your Global Trials?
Stop treating language as a downstream task. Join the clinical leaders who use Language Scientific to scale safely and efficiently.
Clinical Research Translation FAQ's
What types of documents can you translate for clinical research?
Language Scientific translates a wide range of clinical research materials, including clinical trial protocols, informed consent forms (ICFs), investigator brochures, clinical study reports, case report forms (CRFs), and regulatory submissions.
How do you ensure translations meet regulatory requirements?
Language Scientific’s translations comply with global regulatory standards, including FDA, EMA, and ICH guidelines. We work with certified linguists who specialize in clinical research and understand the nuances of regulatory language.
Do you provide back translation and reconciliation services?
Language Scientific offers back translation and reconciliation services to ensure the accuracy and reliability of critical documents, particularly those submitted to regulatory authorities.
How do you maintain confidentiality in clinical translations?
Language Scientific has strict data protection protocols in place, including secure file transfers and non-disclosure agreements (NDAs) with all team members, to safeguard sensitive clinical data.
Can you handle translations for global clinical trials?
Language Scientific’s expert linguists cover over 215 languages, ensuring precise translations for multinational clinical trials. We understand the cultural and linguistic nuances needed for diverse target audiences.
How quickly can you deliver translations?
Language Scientific works with our clients to deliver high-quality, AI-optimized translations on time. Our streamlined workflows and dedicated project managers ensure quick turnaround times without compromising quality. We work with you to meet strict clinical trial deadlines.
What industries do you specialize in for clinical translations?
Language Scientific specializes in life science translation services, including pharmaceuticals, biotechnology, and medical devices, catering to the specific needs of Clinical Research Organizations (CROs).
Can you translate patient-facing documents for clinical trials?
Language Scientific provides culturally appropriate translations for patient-facing materials such as informed consent forms, patient diaries, and questionnaires, ensuring comprehension and compliance.
How do you ensure linguistic accuracy in clinical translations?
Language Scientific linguists are subject-matter experts with clinical and scientific backgrounds. Every project undergoes rigorous quality assurance processes, including editing and proofreading by senior linguists.
Can you handle rush projects?
Language Scientific offers expedited services for urgent projects while maintaining our commitment to quality and accuracy. We work with you to meet tight clinical trial deadlines.